|
Solar Alkaline Battery Charger
By: Dave Johnson
I received an email the other day from a guy who was tired of replacing alkaline
batteries every 2 months in his wireless weather station. He wanted some way to
draw energy from the sun, so he could keep the station running for a much longer
time before the batteries had to be replaced. |
 |
| I wonder if
he could extend the operating time to years by pumping a small amount of current
into the two AAA 1.5v alkaline cells inside the weather station during the day.
The idea is to put a bit more average current into the battery than is taken
out. In theory, this should keep the battery capacity at some constant level,
extending its life to many years. But, would this idea work? |
 |
 |
 |
|
Weather Station |
Alkaline AAA Cells |
Alkaline AAA Cells |
|
| If you divide
a 1 Amp-hour AAA alkaline cell by 60 days, then divide that by 24 hours, you get
a figure of 0.7 millamps. This would be the average current drawn from the two
cells by the weather station circuit. This is a pretty small amount of current
but it is high enough to deplete the AAA cells in just 60 days. Increasing the
battery size to a pair of AA cells might increase the time to 4 months. Going
all the way to two big D cells might result in an operating time of 2 years.
That still may not be long enough for a guy who has to climb up on a roof to
replace those batteries. |
How about
NiMH
rechargeable cells, charged by a solar panel? Yes, this might work but
since the weather station wants to see 3 volts from the battery pack and two
NiMH
cells would only deliver 2.4 volts, three cells would need to be used,
producing 3.6v. That is certainly possible. But, could the weather station
tolerate a voltage of 4 volts during battery charging cycles? Maybe! I just
don’t know. I suppose a good 3v voltage regulator and three NiMH
cells could be
made to work. |
Could a super
capacitor work? Let’s see. Let’s assume that the weather station needs to
operate for two full days under dark conditions, drawing power only from the
capacitor.48 hours times 3600 seconds per hour is 172,800 seconds. If two
2.5v super capacitors were wired in series they could be charged up to 5v. By
using a very good
3v voltage regulator, the capacitor could be discharged down to maybe 2.7v
before the weather station would no longer function properly. This leaves a
voltage change of about 2 volts. The equation for the rate of voltage drop in a
capacitor with a fixed current is dV/dt = I/C where dV/dt is the voltage change
with respect to time, I is the current in Amps and C is the capacitance in
farads. If we plug in the 2 volts change into the equation and 172,800 seconds,
with a current draw of 0.0007 Amps, we get a figure of 60 farads. Since two
capacitors would have to be wired in series, each would have to have a value of
at least 120 farads. Although such capacitors do exist, they would be bulky and
expensive. The charging circuit would also have to have some kind of balancing
circuit across each cap to insure the voltage was not exceeded. The real
advantage of this approach would be a long 10 year life or more.
|
 |
I still like the trickle
charge option. I would like to see someone test the concept of using the
existing alkaline AAA cells installed in the weather station and use a
small solar panel to push a bit of current into the cells. Could this
allow the battery to last 5 years? I think it would be worth a try.
There is not much data
on trickle charging alkaline batteries. Some commercial alkaline battery
chargers exist but they dump a lot more current into the cells than needed
in this application. Battery manufactures say that their batteries could
be explode if charged but again, by limiting the charge current to just a
few milliamps, I don’t see that happening.
I ran a quick test on
a fresh alkaline cell. The open circuit measured 1.5v. With 3ma of
current flowing into the device for several hours, the voltage across the
cell during charging increased to 1.65 volts. With two cells under
charge, the voltage would be 3.3v. I think the weather station would be
able to handle that voltage just fine. |
|
140 Farad Super Capacitors |
|
|
A typical day has about 6
hours of useable sunlight. If the station needs 0.7 milliamps for 24 hours,
then that means about 17 milliamp hours would be drawn from the battery each
day. To make up for that battery discharge, the solar panel would need to pump
about 3 milliamps into the battery and to the weather station load during the
day. In theory, this current would average out to a slightly positive value,
going into the battery every day. |
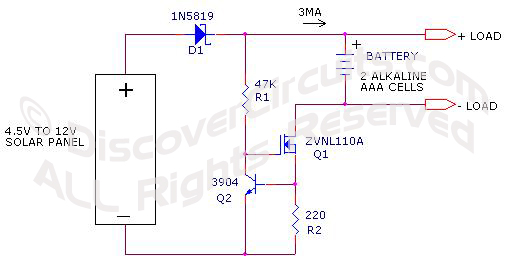
| A 9 or 12
cell solar panel should work well, if the current from the solar panel to the
battery was limited to 3 milliamps. I have drawn up a simple current regulator
which will work with solar panels up to 18v or so. |
 |
 |
|
4.5v Solar Panel |
5.0v Solar Panel |
|
| Even cheap
solar cells have an efficiency of about 10%. During a bright sunny day about
0.7 watts of solar power strikes every square inch of a solar cell. So, with an
efficiency of 10% about 0.07 watts of power should be available for each square
inch. Each solar cell produces a voltage of about 0.5v so with 0.07 watts, each
square inch of solar cell area should be able to crank out 140ma of current.
This is a lot more current than needed for this application. In fact, if the
goal is a current of only 3ma, with a voltage of about 4.5v, the solar panel
area would only need to be about 0.5 inches by 0.5 inches. But, such solar
panels are rare. To produce a voltage of about 4.5v to charge the 3v battery, 9
solar cells in series would be needed. A 5v panel would require 10 solar
cells. The circuit below will certainly work with panels with a higher voltage
if that is all you have. If someone tries this trickle charge method, let me
know how it works out. |
|
 |
|
3ma Current
Limiting Circuit |
|
|